Immune Safety Avatar
Nonclinical mimicking of the immune system effects of immunomodulatory therapies
The imSAVAR Vision
The vision of Immune Safety Avatar (imSAVAR) is to develop a platform for the integrated nonclinical assessments of safety and efficacy of immunomodulatory therapeutic modalities.
The imSAVAR project is a pioneering initiative funded by the Innovative Medicines Initiative (IMI), dedicated to enhancing the nonclinical safety assessment of immunotherapies.
The primary objective is the creation of a platform to significantly improve the prediction of potential side effects and thereby the safety and efficacy of immunomodulatory therapeutics. We want to achieve that by developing fit-for-purpose nonclinical models with high transferability from laboratory settings to real-world applications.
As partners from the private sector, pharma, regulators, and technology providers closely collaborate in this consortium, imSAVAR will benefit the field of immune safety evaluation. Through these concerted efforts, imSAVAR aims to positively impact the field of immune safety evaluation and pave the way for the future of immunomodulatory therapeutics.
Upcoming Events
- No Events

“If we really want to move forward with these modern immunomodulatory therapies we need much more research on predictive markers and predictive models. With the imSAVAR project we aim to tackle the challenge of a better understanding of the complexity of the human immune system.”
Ulrike Köhl / Coordinator

“The imSAVAR team plans to characterise a range of innovative human-relevant models and biomarkers to enhance translational safety assessment of immunomodulatory therapeutics and ultimately lead to the development of safer efficacious medicines for patients.”
Jonathan Moggs / Coordinator
Learn more about the imSAVAR project
Concept
Read more about our challenges, approach, objectives and the impact we want to make.
Consortium
The interdisciplinary imSAVAR consortium consists of 28 international partners from 11 nations.
Forum
Engage in lively discussions and help built up a strong imSAVAR stakeholder community.
Learn more about the imSAVAR project
Concept
Read more about our challenges, approach, objectives and the impact we want to make.
Consortium
The interdisciplinary imSAVAR consortium consists of 28 international partners from 11 nations.
Latest News
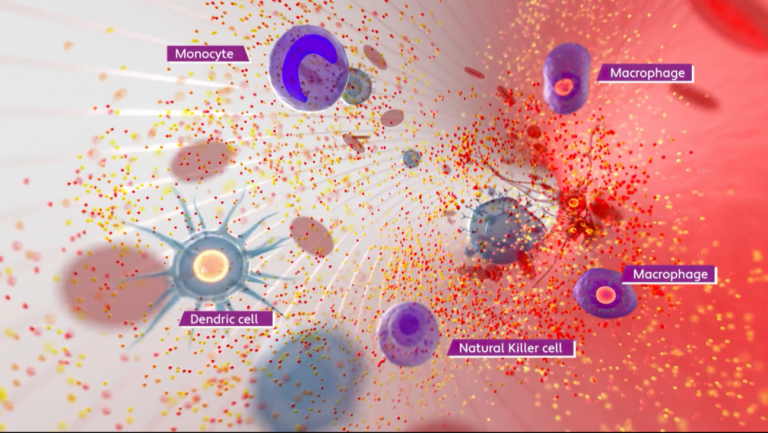
New imSAVAR video released: Understanding immune safety through Adverse Outcome Pathways
A newly released explainer video introduces Adverse Outcome Pathways (AOPs) as a concept to better understand immune-related adverse effects and improve the prediction of drug safety. The video explains

New imSAVAR Video Released: Exploring Organ‑on‑Chip – A Game Changer for Immunotherapies
We are delighted to announce the public release of our new Organ‑on‑Chip explainer video, now available in the project publications section. This video highlights one of the most innovative and
7th imSAVAR Consortium Meeting – November 2025
Final imSAVAR Meeting in Brussels – November 12–13, 2025 On November 12 and 13, the imSAVAR consortium gathered for its final meeting at the Fraunhofer EU Office in Brussels.